Let me explain this a little more. In my Organic Chemistry class, we discussed shapes of molecules, and my professor gave us the example of a molecule found in both oranges and lemons. Both molecules have the formula C10H16 (or CH3C6H8CH2CH3 in the expanded formula). The only difference between the molecules is that the groups found at the bottom of the carbon rings are in different positions. Here's a graphic to explain that a little better:
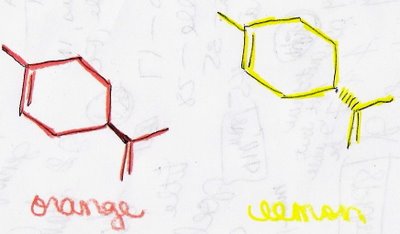
If you can't tell, the groups are pointed in different directions. You have to imagine this in 3-D, which let me tell you can be quite difficult. The group on the orange molecule is coming out of the picture, as referenced by the dark line. Conversely, the group on the lemon molecule is drawn back into the picture, as referenced by the dashed line. Now does that make a little bit more sense???
Now, the main part of this lesson was to explain why oranges and lemons taste different to us when we eat them (or drink their juice). The enzymes and taste receptors in the body are chiral, meaning they are not symmetrical and read things in a certain direction. Because these molecules are also chiral, they are read differently by the taste receptors and enzymes in the mouth. The fact that the bottom groups are in different directions gives the molecules different tastes. This also shows that even changing the shape of a molecule can dramatically alter it's properties, in this case, taste.
So, there's your Organic Chemistry lesson for today. I hope at least part of that was interesting to you, since I really thought that was cool. But anyways... Thank you to everyone out there that has sent in their survey responses. I have gotten almost 100% participation only one day after sending it out, so that is very good. I thank you...
Well that's really all I wanted to share tonight. If I can think of anything else to post about, I'm sure I will have ample opportunities to post this weekend. Enjoy!
 (Hint: Look in the lower left-hand corner.)
(Hint: Look in the lower left-hand corner.)